Applied Cells is pleased to announce a collaboration with SHENTEK to integrate fast QC release solution with Applied Cells’ GoFast™ CAR-T manufacturing workflow on the MARS® platforms.
GoFast™ CAR-T is Applied Cells’ advanced rapid CAR-T manufacturing workflow designed to support closed-fluidic CAR-T production in less than 72 hours. Built on the MARS® platforms, the workflow simplifies key steps including immunomagnetic T cell isolation, activation and transduction, wash, concentration, and media exchange in a streamlined process. GoFast™ workflow is designed to help cell therapy teams reduce complexity, accelerate manufacturing timelines, and support more scalable and cost-effective CAR-T production.

Through this collaboration, Applied Cells and SHENTEK will work together to launch Hubble™, a qPCR platform with fast QC capabilities that supports same-day QC release testing of cell therapy products.
Hubble™ provides a hands-free approach by combining nucleic acid extraction and real-time PCR into a fully integrated, automated single-use cartridge solution. This system is designed to deliver results from sample-to-result in less than 3 hours, and offers walk-away operation. The platform features integrated internal controls, 21 CFR Part 11 compliance, and broad detection coverage for more than 180 species of Mycoplasma, Spiroplasma, and Acholeplasma currently, with sterility, VCN, and RCL capabilities planned for the near future.
“Fast, reliable QC is essential to making rapid CAR-T manufacturing more accessible,” said Yuchen Zhou, CEO of Applied Cells. “By collaborating with SHENTEK, we are providing the cell therapy industry a more complete, even faster, and further lower-cost solution. Together, we aim to help cell therapies move closer to affordability, and scalable production.”
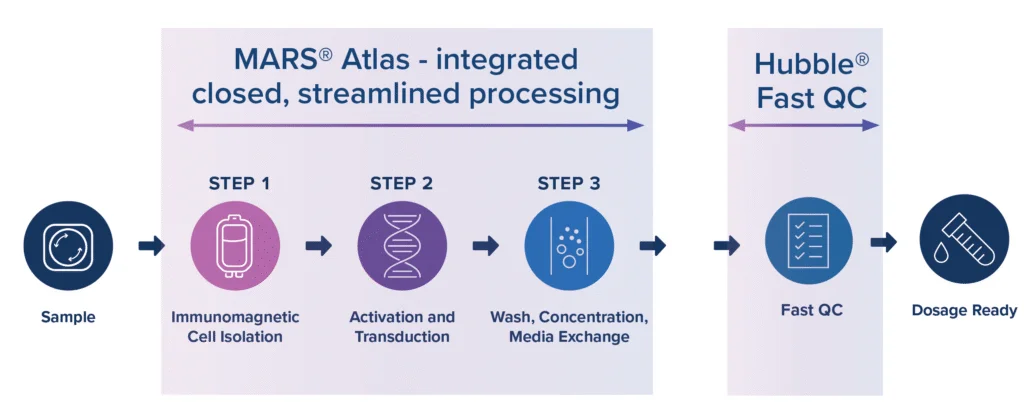
This collaboration is part of Applied Cells’ broader mission to enable low cost, fast, simple, and scalable cell therapy manufacturing for global users. By pairing the MARS® GoFast™ workflow with Hubble™, Applied Cells and SHENTEK aim to accelerate patient access of life-saving CAR-T cell therapies around the world.
Applied Cells and SHENTEK will showcase the GoFast™ CAR-T workflow and Hubble™ Fast QC capabilities together at ASGCT 2026. Attendees can visit Poster #2372 for GoFast™ CAR-T, Poster #1197 for Hubble™ Fast QC, and Booth #932 to learn more about the collaboration and early access opportunities.
For more information, visit Applied Cells at ASGCT Booth #932 or contact info@appliedcells.com.
About Applied Cells
Applied Cells, Inc. develops advanced cell separation, enrichment, and processing technologies designed to support cell therapy, tumor biology, and cell and nuclei enrichment workflows. The company’s MARS® platforms are built to help researchers and therapy developers streamline complex cell processing workflows with scalable, high-quality technologies.
About SHENTEK
SHENTEK develops automated testing solutions for bioprocessing and advanced therapy workflows. Its AdvSHENTEK Mycoplasma DetectInnova System is designed to provide rapid, automated, and easy-to-use mycoplasma detection for biotherapeutic and cell therapy applications.
Kat Ginda-Mäkelä
For research use only. Not for use in therapeutic or diagnostic procedures.

